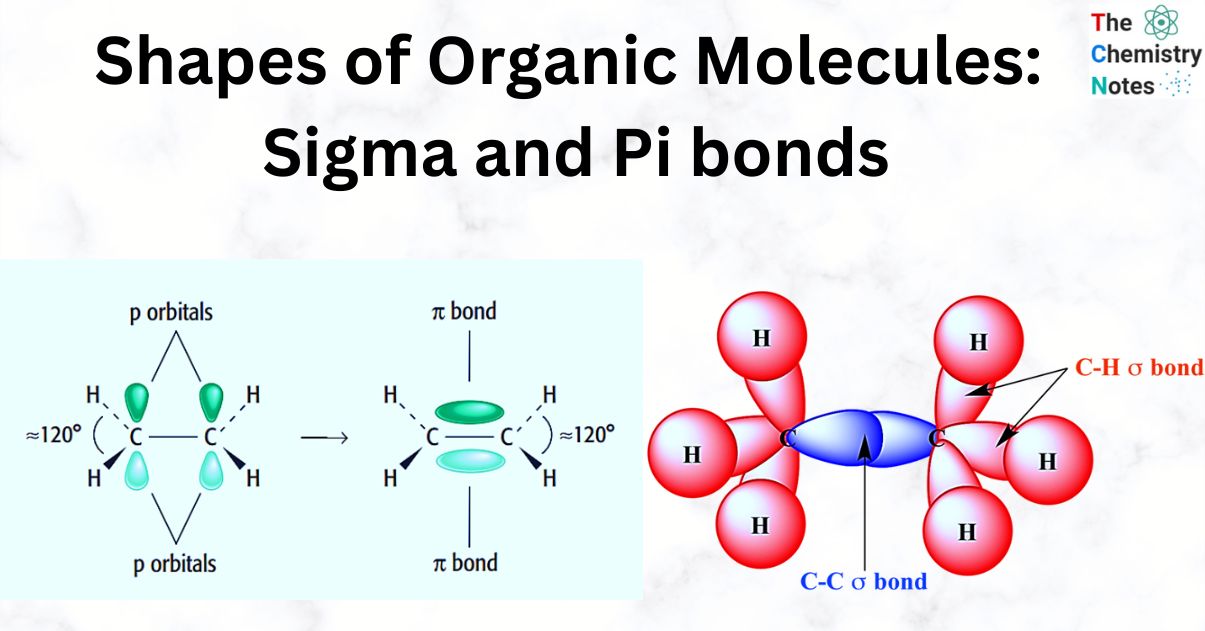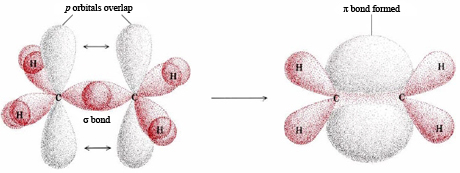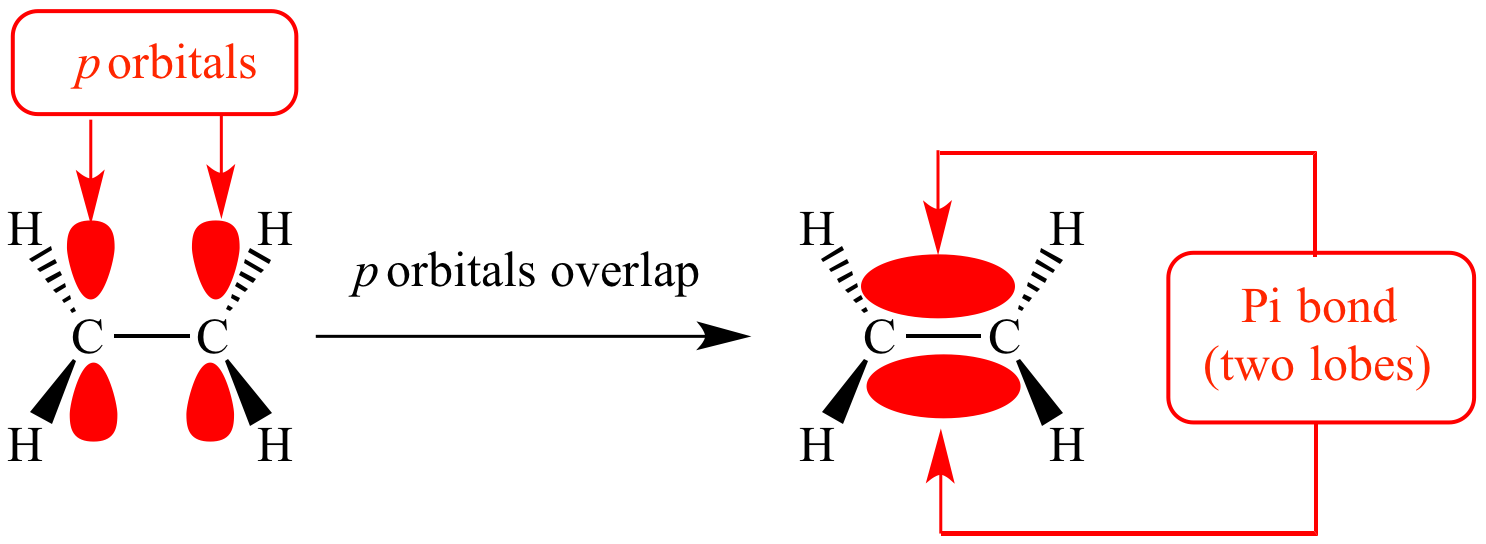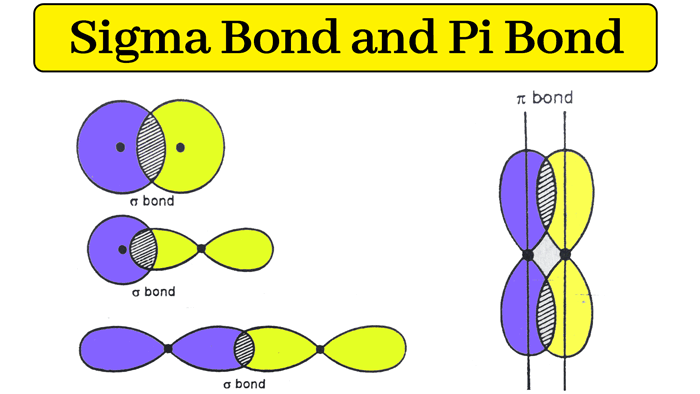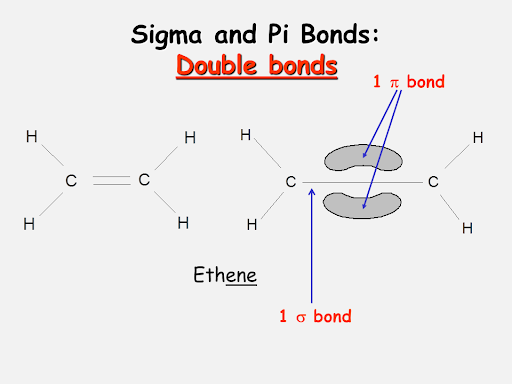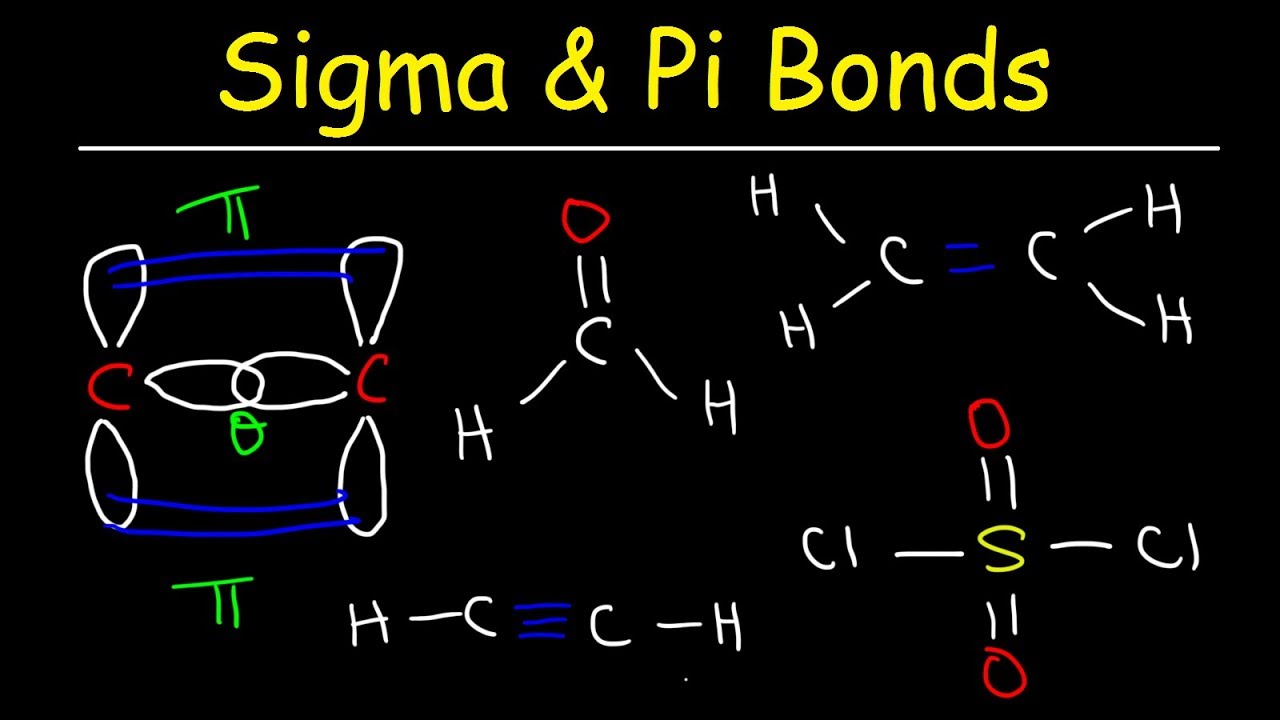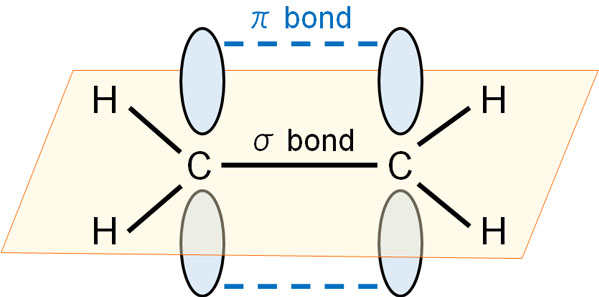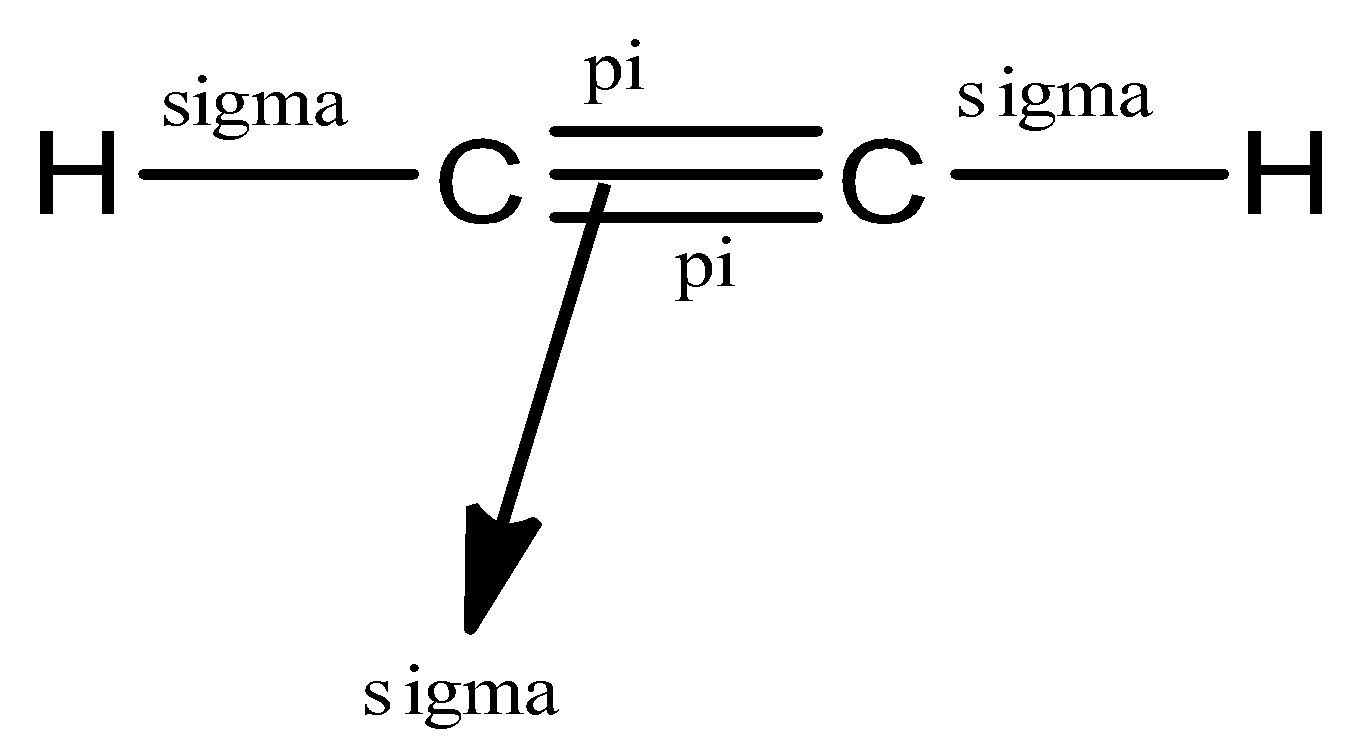
The total number of sigma and pi bonds in the following molecule?(a)- ${{C}_{2}}{{H}_{2}}$ (b)- ${{C}_{2}}{{H}_{4}}$
How Many Sigma and Pi Bond (Count Number of Sigma and Pi Bonds) Example, Practice Problem, Shortcut - YouTube

A double bond consists of: 1. two sigma bonds. 2. two pi bonds. 3. an alpha and a beta bond. 4. two beta bonds. 5. one sigma bond containing four electrons. | Homework.Study.com